NGS in Veterinary Oncology: What's Commercially Available Now and Where It's Headed
If you've been following developments in human oncology over the past decade, you'll be familiar with the concept of tumor molecular profiling — sequencing a patient's cancer to identify actionable mutations and guide targeted therapy. The logical question is: how much of that is available for our patients?
The short answer is that next-generation sequencing (NGS) has arrived in veterinary medicine, but it remains in early clinical adoption, the available assays are almost exclusively validated for dogs, and these tools work best as an adjunct to histopathology rather than a replacement for it. This post offers an honest overview of where things stand today — what you can actually order, what the evidence shows, and where the field is likely headed.
What is NGS and Why Does It Matter in Oncology?
Next-generation sequencing refers to high-throughput DNA sequencing technologies capable of reading millions of short DNA fragments in parallel. In oncology, the primary application is somatic mutation profiling — sequencing tumor DNA to identify single-nucleotide variants (SNVs), insertions and deletions (indels), copy number alterations (CNAs), and gene fusions that may be driving neoplastic behavior.
In human medicine, this information is routinely used for three purposes: diagnostic clarification in poorly differentiated or ambiguous tumors, prognostic stratification, and selection of targeted therapies. Veterinary medicine is beginning to develop a parallel framework, though the evidence base is considerably younger and the therapeutic options are more limited.
What's Commercially Available in the USA Right Now?
Two tissue-based NGS panels are currently available for clinical use in dogs in the United States:
FidoCure® (One Health Company)
FidoCure performs tumor sequencing on FFPE tissue, identifying mutations in a targeted panel of cancer-associated canine genes. The company provides a DNA report to the clinician and facilitates access to FDA-approved oral targeted therapies based on the mutation findings. Turnaround is approximately 2–3 weeks. Independent research validating the test's clinical utility is ongoing.
SearchLight DNA™ (Vidium Animal Health / Antech)
SearchLight DNA evaluates up to 120 cancer-associated canine genes from FFPE tissue, fine needle aspirates, or effusion samples. Mutations are annotated for diagnostic, prognostic, and therapeutic significance. A 2023 study published in JAVMA evaluated SearchLight DNA in 69 dogs with diagnostically ambiguous cancers and found the assay provided clinically useful information — diagnostic, prognostic, or therapeutic — in 86% of cases, with diagnostic clarity achieved in 54% of cases where histopathology alone had not reached a definitive conclusion.
This last point is worth emphasizing for practicing pathologists and oncologists: in diagnostically challenging cases — poorly differentiated neoplasms, ambiguous round cell tumors, histologically atypical presentations — mutation profiling may provide meaningful supplemental information when morphology and immunohistochemistry have reached their limits.
Quick Reference: Tissue-Based NGS Panels (Dogs)
Summary of current NGS tests available.
The Important Caveat: Validation Is Still Maturing
Both FidoCure and SearchLight DNA are commercially available and in active clinical use — but it's important to be transparent with clients and referring clinicians that independent validation of these assays is ongoing. The significance of all identified mutations is not yet fully characterized, and the evidence base linking specific canine tumor mutations to treatment response is considerably thinner than in human precision oncology.
This isn't a reason to dismiss these tools; it's a reason to use them thoughtfully, with appropriate client communication about what is and isn't known. For oncologists managing cases where conventional therapy has failed or where a definitive diagnosis remains elusive, these panels offer a reasonable next step.
What About Liquid Biopsy?
Liquid biopsy — detection of tumor-derived cell-free DNA (cfDNA) from peripheral blood — represents a separate and complementary application of NGS. OncoK9 (PetDx) was the first commercially available canine liquid biopsy test, with published data showing a sensitivity of approximately 55–62% and specificity of approximately 97–98% across 30 canine cancer types. However, PetDx appears to have discontinued service as of 2024.
In January 2025, IDEXX Laboratories launched IDEXX Cancer Dx, a blood-based test targeting early canine lymphoma detection with a reported 2–3 day turnaround. The liquid biopsy space continues to evolve rapidly, and it's reasonable to expect new entrants and improved sensitivity as the field matures.
It is worth noting that liquid biopsy and tissue-based NGS serve different diagnostic questions. Liquid biopsy is positioned primarily as a screening or monitoring tool; tissue-based sequencing remains more appropriate for mutation profiling in the context of an established diagnosis.
The Gap in Feline and Exotic Medicine
One of the most significant limitations in the current landscape is the near-complete absence of validated NGS tools for species other than dogs. There are no commercially available multi-gene cancer panels for cats, birds, rabbits, ferrets, or other exotic companion animals. Some single-gene PCR assays exist — notably KIT mutation testing for feline injection-site sarcomas and mast cell tumors — but comprehensive tumor mutation profiling in these species remains a research-stage endeavor.
For those of us working with non-canine species, this gap is a real one. Histopathology, immunohistochemistry, and single-gene assays remain the standard of care, and will continue to be the primary diagnostic tools for the foreseeable future.
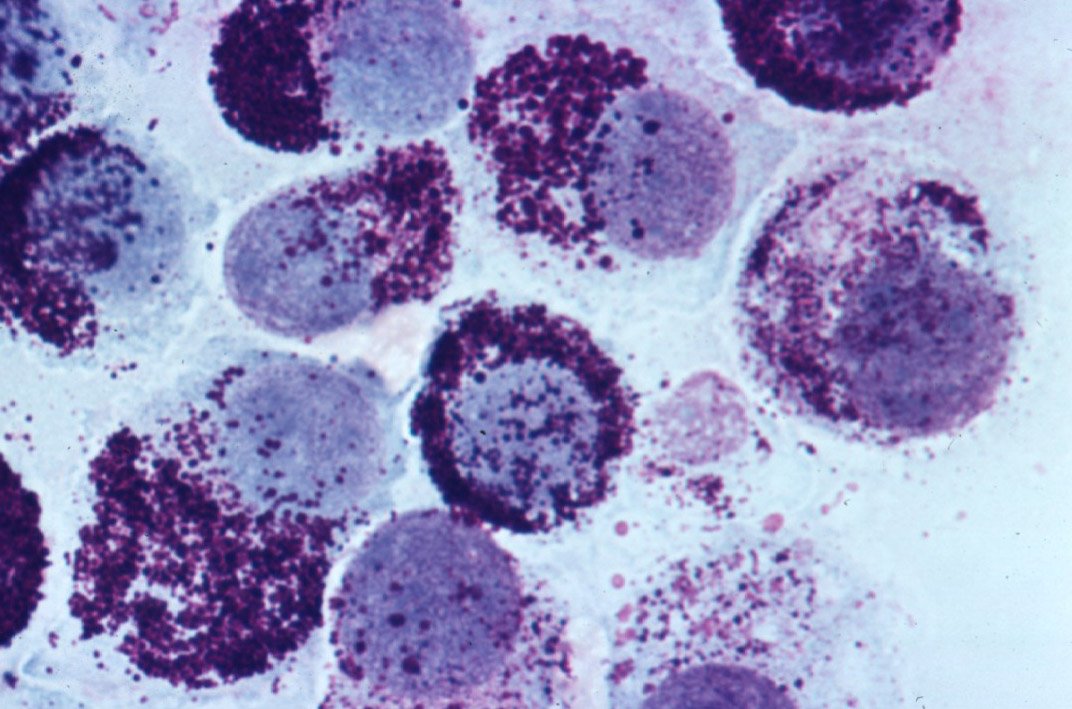
Mast cell tumor on cytology. NGS testing on these tumors is expected to increase with time, offering more precise prediction of behavior.
The Relationship Between NGS and Histopathology
Perhaps the most important takeaway from the current state of veterinary NGS is that these tools are designed to build on histopathology, not to replace it. The workflow for both FidoCure and SearchLight DNA begins with a tissue biopsy, which is typically reviewed by a pathologist before or concurrently with molecular testing. The mutation report is then interpreted in the context of the histologic diagnosis.
This is the same paradigm used in human precision oncology — morphology and immunohistochemistry establish the diagnosis; molecular profiling refines it and guides therapy. There is no scenario in which a mutation report substitutes for a careful histopathologic evaluation. The two are complementary.
Where Is This Heading?
The veterinary oncology diagnostics market is growing, with investment coming from both established diagnostic companies and biotech startups. Several trends are worth watching:
• Expanded species coverage: As the canine genomic landscape becomes better characterized, feline and exotic animal oncogenomics will likely follow, driven in part by the comparative oncology community.
• Improved liquid biopsy sensitivity: Current cfDNA assays are limited by sensitivity, particularly for solid tumors with low circulating tumor DNA. Improved library preparation and sequencing depth will address this over time.
• AI-assisted mutation interpretation: Machine learning approaches to annotating veterinary-specific mutations against canine and comparative oncology databases will improve the clinical interpretability of sequencing reports.
• Integration with digital pathology: The convergence of whole slide imaging and tumor genomics — where morphologic and molecular data are interpreted together — is an active area of development in both human and veterinary medicine.
The Bottom Line
NGS-based tumor profiling is a real and clinically available tool for dogs — not science fiction, but also not yet routine practice. For oncologists managing canine cases, these assays offer a reasonable adjunct in diagnostically ambiguous cases or when mutation-guided therapy is being considered. For clinicians working with cats and exotic species, validated multi-gene panels do not yet exist, and the standard histopathologic workup remains essential.
As these tools develop, histopathology will remain the diagnostic foundation. Molecular profiling adds a layer of information; it does not replace the interpretive work that begins at the microscope.
About the Author
Eric Snook, DVM, PhD, DACVP is a board-certified veterinary pathologist and founder of Vetopathy, an independent histopathology service. He holds a PhD in neurodegenerative diseases with post-doctoral training in neuromuscular disease and brings over 20 years of diagnostic experience to private practice. Vetopathy offers sub-48-hour turnaround, direct pathologist access, and pre-submission guidance for veterinary specialists and clinics across the USA.