Feline Injection-Site Sarcoma: Revisiting Pathogenesis in the Light of Current Evidence
Feline injection-site sarcoma (FISS) is not a new diagnosis. It has been recognized since the early 1990s, when pathologists began noticing an unusual cluster of aggressive subcutaneous sarcomas — predominantly fibrosarcomas — developing at sites commonly used for vaccination in cats. The association was striking enough that a dedicated task force was formed in the United States in 1997, vaccination site recommendations were overhauled, and the condition has since become one of the most studied adverse events in veterinary medicine.
And yet, more than three decades later, the pathogenesis of FISS remains incompletely understood. The broad strokes are established: chronic local inflammation at an injection site appears to trigger malignant transformation of fibroblasts or myofibroblasts in genetically susceptible cats. But the molecular details — what drives that transformation, why only certain cats develop it, and which specific pathways sustain tumor growth — are still being worked out. Recent years have brought new insights from transcriptomic analysis, tumor microenvironment research, and molecular profiling that are beginning to fill in those gaps, and have important implications for diagnosis, histologic interpretation, and emerging therapeutic strategies.
This post summarizes where the science currently stands.
What We Know: The Inflammation-Transformation Hypothesis
The most widely accepted model frames FISS as a consequence of a dysregulated local inflammatory response. Injected materials — vaccine adjuvants (particularly aluminum hydroxide), long-acting corticosteroids, microchips, suture material, and other foreign bodies — trigger a chronic granulomatous or lymphoplasmacytic inflammatory reaction in the subcutis. In most cats, this reaction resolves without incident. In a small subset, the sustained inflammatory milieu appears to drive progressive genetic instability in the local fibroblast and myofibroblast population, eventually giving rise to neoplastic transformation.
This model explains several clinical observations: why the sarcomas cluster at injection sites, why they are histologically distinct from non-injection-site fibrosarcomas (showing greater pleomorphism, higher mitotic indices, and characteristic peritumoral lymphoplasmacytic inflammation), and why adjuvanted vaccines — which generate more pronounced and longer-lasting local inflammation than non-adjuvanted formulations — appear to carry a higher associated risk.
Recent CT imaging data quantified this difference directly: in cats receiving adjuvanted vaccines, mean inflamed tissue volume on day 7 post-injection was nearly 300 times larger than in cats receiving non-adjuvanted vaccines (Haist et al., 2023). Whether this inflammatory magnitude difference translates directly to a proportional difference in FISS risk has not been definitively established.
Importantly, even non-adjuvanted vaccines are not risk-free. The chronic inflammatory response appears to be necessary but not sufficient for malignant transformation — individual host genetics play a significant role in determining which cats progress from inflammation to neoplasia.
The Molecular Layer: PDGF, TP53, and Transcriptomic Findings
The chronic inflammation hypothesis tells us what the trigger is, but not the mechanism of transformation. Several lines of molecular evidence have begun to map that terrain.
PDGF and receptor tyrosine kinase signaling. Platelet-derived growth factor (PDGF) and its receptor (PDGFR) are consistently overexpressed in FISS and are largely absent from non-injection-site fibrosarcomas — a distinction that has been recognized for some time but whose therapeutic implications are now being explored more seriously. PDGF promotes fibroblast proliferation and survival, and PDGFR overexpression likely contributes to unchecked cell division in established tumors. This is the molecular basis for interest in tyrosine kinase inhibitors such as imatinib in FISS, with early phase 1 data showing short-term stabilization in a subset of treated cats.
TP53 mutation. Loss of functional p53 is a recurring finding in FISS and fits the model well: p53 is a critical guardian of genomic integrity, and its inactivation removes a key checkpoint that would ordinarily trigger apoptosis in cells accumulating DNA damage during chronic inflammation.
PI3K/mTOR pathway dysregulation. A landmark transcriptomic study (Gould et al., 2019) performed the first mRNA sequencing of FISS tumors compared to normal feline skin and found that the PI3K and mTOR signaling pathways were among the most significantly upregulated in FISS. The authors specifically identified dual targeting of PI3K and mTOR as a potentially actionable therapeutic approach. Importantly, the FISS transcriptome showed strong similarity to soft-tissue sarcomas in dogs and humans, suggesting that findings from comparative oncology may translate meaningfully across species.
Inflammation, Ki-67, and tumor progression. A 2023 study evaluating metallothionein expression in FISS demonstrated a correlation between the Ki-67 proliferative index and inflammatory score, suggesting that inflammation is not just a pathogenic trigger but continues to play an active role in tumor progression (Mikiewicz et al., 2023). The inflammatory infiltrate may be sustaining the proliferative drive of established tumor cells rather than simply preceding their development.
Matrix metalloproteinases and invasiveness. MMP-2 and MMP-9 are variably expressed in FISS and contribute to extracellular matrix remodeling and the tumor's characteristic invasiveness along fascial planes. While their expression patterns have not proven to be reliable individual prognostic markers, they are part of the molecular machinery that makes FISS so difficult to excise with clean margins.
The Tumor Microenvironment: Macrophages as Promoters
One of the more interesting emerging threads in FISS research concerns the role of tumor-associated macrophages (TAMs). The inflammatory infiltrate characteristic of FISS — the peritumoral lymphoplasmacytic cuff that pathologists recognize as a hallmark feature — is not simply a bystander response. TAMs within that microenvironment may actively promote tumor progression by enhancing angiogenesis, increasing tumor cell motility and invasiveness, and suppressing anti-tumor immune responses.
This is an area of active investigation. Research specifically targeting TAMs in feline FISS is early stage, but the parallels with human soft-tissue sarcoma biology are striking and suggest that immunomodulatory strategies — including potential future checkpoint inhibitor approaches — may eventually be relevant in FISS management. For now, this remains a research frontier rather than a clinical tool, but it is worth tracking.
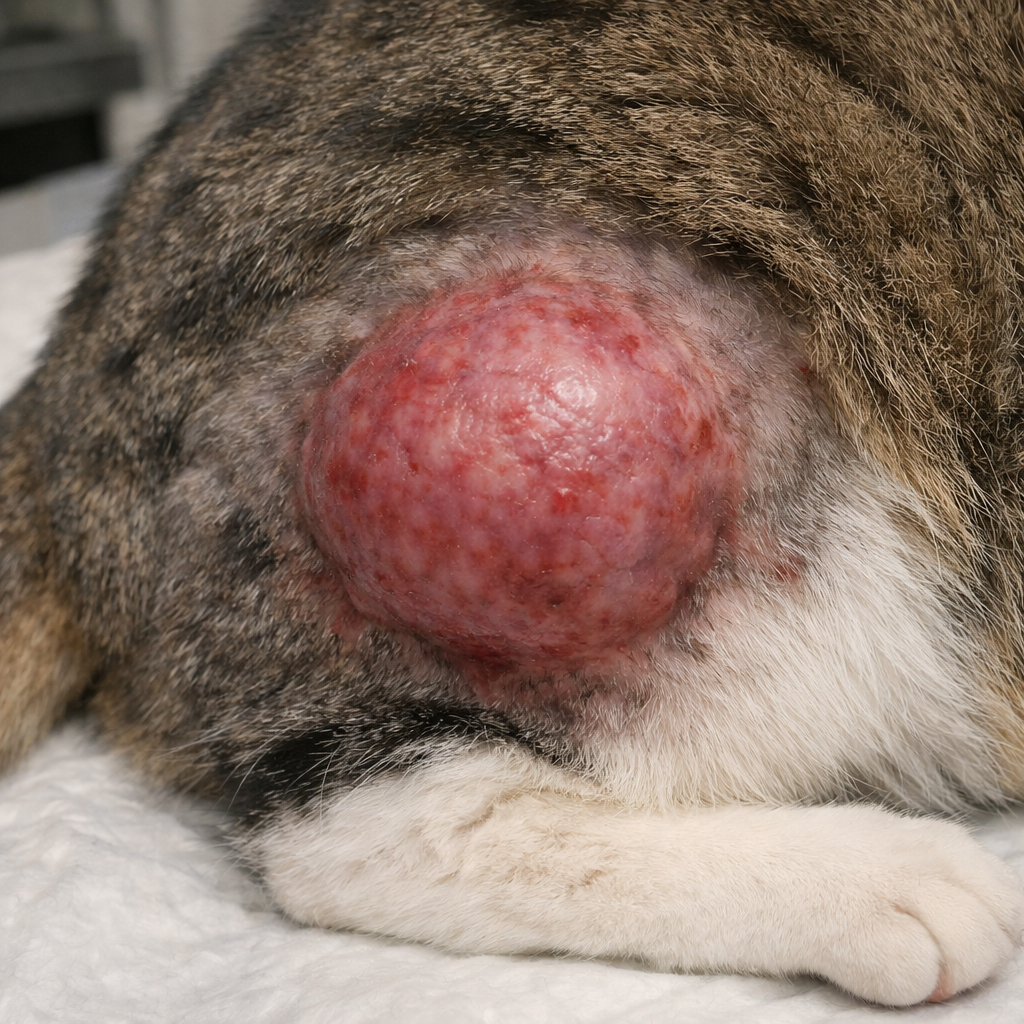
Possible injection site sarcoma on the right rear limb of a cat. It is recommended that vaccines and injections be given as distal in the limb as possible to allow for amputation and complete excision, if necessary.
What This Means for Histologic Interpretation
Understanding the molecular pathogenesis of FISS shapes how we should interpret these tumors at the microscopic level and what information a pathology report should communicate.
The diagnosis itself is primarily histomorphologic and contextual: a pleomorphic spindle cell sarcoma (most commonly fibrosarcoma) arising in the subcutis at a known injection site, with characteristic peritumoral lymphoplasmacytic inflammation, central necrosis, and high mitotic activity. Immunohistochemistry is used to confirm mesenchymal lineage and exclude other differential diagnoses (carcinoma, lymphoma, histiocytic sarcoma) rather than to establish the FISS diagnosis per se. Notably, no validated histologic grading system specific to FISS has been established. While general soft tissue sarcoma grading criteria have been applied in some studies, they have not demonstrated consistent prognostic correlation in this tumor type. Surgical margin status remains the most reliable histopathologic prognostic indicator.
From a prognostic standpoint, the features that consistently matter are tumor size (with an inflection point around 3.5–4 cm), mitotic count (with counts above 20/10 HPF associated with higher recurrence risk), and — critically — surgical margin status. The disease-free interval in cats with tumor-free margins can extend to approximately two years; with incomplete margins, recurrence within 100 days is common. This makes accurate margin assessment one of the most clinically impactful elements of the histopathology report.
A practical note on submission: if your surgeon suspects or confirms FISS, orient the specimen before fixation and communicate the anatomic margins clearly on the submission form. Margin adequacy is the single most important prognostic variable the pathologist can report, and it is only as accurate as the information provided about specimen orientation.
The Adjuvant Question: Unresolved but Practically Important
Despite decades of investigation, the question of whether adjuvanted vaccines carry a meaningfully higher FISS risk than non-adjuvanted formulations remains contested in the literature. Several studies have found no statistically significant difference in risk; others have suggested a higher risk with adjuvanted products. The 2023 ABCD guideline update (Hartmann et al., Viruses 2023) takes the position that where non-adjuvanted vaccines of equivalent efficacy are available, they are a reasonable preference pending better mechanistic understanding — but that where adjuvanted vaccines offer superior protection for high-risk cats, that clinical benefit should not be abandoned over theoretical FISS risk.
What is not contested: the incidence of FISS in vaccinated cats in the USA is estimated at 1–4 per 10,000 vaccinated animals, and vaccination remains one of the most effective preventive health interventions available. The risk-benefit calculation favors vaccination in virtually every clinical scenario. The pathogenesis discussion is relevant not as a reason to avoid vaccines, but as context for understanding what these tumors are, why they behave the way they do, and how to counsel clients who receive this diagnosis.
What Remains Unknown
It is worth being explicit about where the gaps are, because honest uncertainty is more useful to the clinician than false confidence:
• Why only some cats develop FISS after injection-site inflammation is not understood. Individual host genetic susceptibility is assumed but the specific variants involved have not been characterized.
• Whether the observed molecular alterations (PDGFR overexpression, TP53 loss, PI3K/mTOR activation) are causative drivers or passenger events in the transformation process has not been fully resolved.
• The role of the tumor immune microenvironment — particularly TAMs — in established FISS is an active area of investigation but not yet clinically actionable.
• There is no validated serum or tissue biomarker that predicts which cats with injection-site inflammation will progress to sarcoma.
The Takeaway
FISS is best understood as the result of a multistep carcinogenesis process: a chronic inflammatory trigger at a susceptible tissue site, acting on an individual cat with a genetic background that impairs the normal cellular response to DNA damage, leading to progressive molecular dysregulation of fibroblast growth control. The inflammation is not incidental — it is mechanistically central, and it continues to shape the tumor's biology even after establishment.
For the clinician receiving a FISS diagnosis, the key practical points are: aggressive surgery with the widest achievable margins remains the cornerstone of treatment, margin status is the most important prognostic variable, and multimodal therapy (surgery plus radiation, with or without chemotherapy) offers the best disease-free interval. Emerging data on tyrosine kinase inhibitors and immunomodulation are worth following, but are not yet ready for routine use.
For the pathologist, FISS is a diagnosis where the report should go beyond histologic classification to provide the clinician with the information they actually need to make treatment decisions: margin status, mitotic count, depth of invasion, and a clear statement of whether the morphologic and clinical features are consistent with an injection-site versus non-injection-site origin. Formal histologic grading should not be reported for FISS given the absence of a validated species- and tumor-specific grading scheme and the lack of consistent correlation between borrowed grading criteria and clinical outcome in this tumor type.