Encephalitozoon cuniculi in Rabbits: A Pathogen Your Patients Are Likely Already Carrying
Encephalitozoon cuniculi is not a rare or exotic diagnosis. Worldwide seroprevalence in pet rabbits has been reported to range from 41 to 85% depending on geographic region and testing methodology, with most infected animals remaining subclinical throughout their lives. The gap between serologic prevalence and clinical disease is one of the defining features of this pathogen — and one of the central interpretive challenges when managing a rabbit with neurologic, renal, or ocular signs.
For the veterinary clinician, E. cuniculi sits in a diagnostically uncomfortable position: it is common enough to be a reflex differential in almost any sick rabbit, but the available antemortem diagnostics are imprecise enough that a positive titer is often adjunctive rather than confirmatory. Understanding the pathology underlying clinical presentations — and what histopathology can and cannot contribute — is essential for navigating these cases well.
The organism and its transmission
Encephalitozoon cuniculi is an obligate intracellular microsporidian, now classified within the fungal kingdom based on molecular phylogenetic data, despite lacking many features of classical fungi. It infects a wide range of mammalian hosts — rabbits, rodents, carnivores, non-human primates, and immunocompromised humans — but Oryctolagus cuniculus is its primary reservoir host.
Horizontal transmission occurs primarily via ingestion or inhalation of spores shed in the urine of infected animals. Spores are environmentally hardy and can remain viable for weeks under appropriate conditions. Vertical transmission also occurs — transplacental infection during fetal development results in spore deposition in the developing ocular lens, which is the pathogenetic basis for the phacoclastic uveitis seen in young rabbits. This prenatal route partly explains why seroprevalence is detectable even in young animals with limited environmental exposure.
Following ingestion, spores invade intestinal epithelium and are disseminated systemically via infected macrophages. Organs with high blood flow — kidneys, brain, lungs, and liver — are the primary targets. The intracellular location of the organism allows it to partially evade humoral immunity, which is why serologic titers do not reliably correlate with disease activity or severity.
Pathology: what the lesions look like and where
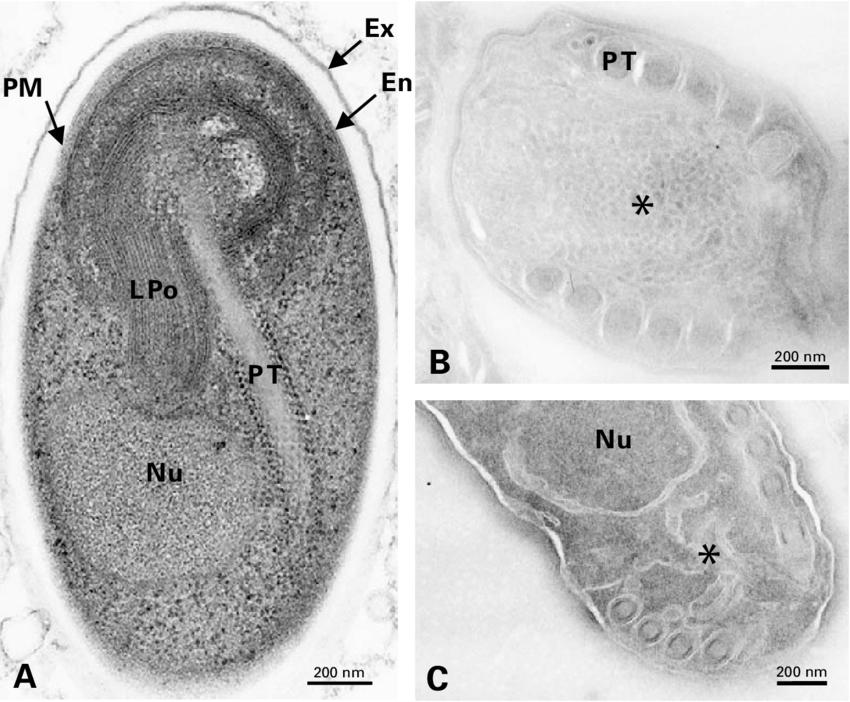
The histopathologic hallmark of E. cuniculi infection is granulomatous to pyogranulomatous inflammation centered on clusters of spores within host cells. Spores measure approximately 1.5 x 2.5 microns — at the lower limit of light microscopic resolution — and stain poorly on routine H&E. Gram stain (E. cuniculi is Gram-positive) and modified trichrome stain are the most reliable light microscopic options for spore identification. Calcofluor white under UV fluorescence is sensitive but requires appropriate microscopy equipment. PCR on formalin-fixed paraffin-embedded tissue is the most specific confirmatory method when spore identification is equivocal.
In the kidney, the classic lesion is multifocal interstitial nephritis with granuloma formation, often accompanied by tubular degeneration and interstitial fibrosis in chronic cases. The cortical surface of affected kidneys may show irregular pitting on gross examination — a finding that, while not pathognomonic, should raise E. cuniculi on the differential list at necropsy. Renal lesions are often the most consistently identifiable finding on postmortem examination, even in rabbits that presented with primarily neurologic signs.
In the central nervous system, lesions are multifocal and distributed through the cerebrum, cerebellum, and brainstem, with a predilection for the cerebrum and leptomeninges. Granulomatous encephalitis and meningoencephalitis are the characteristic patterns. An important and frequently cited observation is that the location of CNS lesions does not reliably correlate with clinical signs — a rabbit with prominent vestibular dysfunction may show minimal histologic change in the vestibular nuclei, while extensive granulomatous lesions are found elsewhere. This dissociation complicates both antemortem diagnosis and postmortem interpretation.
Ocular lesions result primarily from vertical transmission. Spores deposited in the developing lens during fetal development trigger a zonal granulomatous lens-induced uveitis following spontaneous anterior lens capsule rupture — the characteristic phacoclastic uveitis of E. cuniculi. Affected animals are typically young, unilaterally affected, and otherwise systemically healthy. Lens material and inflammatory debris in the anterior chamber, often with a white intraocular mass visible on external examination, is the typical presentation. Histopathology of enucleated globes shows lens capsule disruption, lens-induced uveitis, and — on careful examination with appropriate stains — spores within lens epithelial cells or the inflammatory infiltrate.
The serologic diagnosis problem
The most commonly used antemortem diagnostic is serology — IgG or IgM ELISA — and its limitations are worth stating plainly. A positive IgG titer indicates prior exposure, not active disease. Given that seroprevalence in clinically healthy pet rabbit populations in Europe and North America has been reported between 20 and 68% depending on the study, a positive titer in a rabbit with neurologic signs is consistent with E. cuniculi disease but does not confirm it. Otitis media/interna, Pasteurella multocida, Listeria monocytogenes, and trauma are among the differentials for vestibular disease that must be considered regardless of serologic status.
IgM titers have been proposed as markers of more recent or active infection, with the reasoning that IgM elevations precede and are shorter-lived than IgG responses. In practice, the utility of IgM as a disease activity marker remains debated, and serial titers showing rising IgG are probably more informative than a single measurement. The honest framing for clients and clinicians: serology supports or is consistent with E. cuniculi as a diagnosis; it rarely confirms it in isolation.
PCR on urine, cerebrospinal fluid, or tissue samples offers higher specificity than serology but has its own limitations — intermittent shedding reduces urine PCR sensitivity, and CSF collection carries procedural risk in a neurologically compromised rabbit. At necropsy, PCR on formalin-fixed tissue combined with histopathology and special stains provides the most diagnostically complete picture.
Electron microscopic image of E. cuniculi spores.
Vanessa Taupin. La diff´erenciation sporale chez les microsporidies : imagerie 3D et isolement des stades de d´eveloppement, analyse de l’expression diff´erentielle de prot´eines structurales et premi`ere identification des glycanes. Genetics. Universit´e Blaise Pascal - Clermont-Ferrand II, 2006. French
Clinical presentations and their pathologic correlates
Three clinical syndromes dominate E. cuniculi presentations in pet rabbits, each with a distinct pathologic substrate.
Vestibular disease — head tilt, ataxia, nystagmus, rolling — is the most commonly reported presentation and the one most often attributed to E. cuniculi in clinical practice. The underlying pathology is granulomatous encephalitis affecting vestibular pathways, though as noted above, the correlation between lesion location and clinical signs is imperfect. Otitis media/interna must be excluded before attributing vestibular signs to E. cuniculi, as the treatments differ and otitis is both common and treatable.
Renal disease presents as polyuria, polydipsia, weight loss, and progressive azotemia. The pathologic basis is chronic granulomatous interstitial nephritis with fibrosis and nephron loss. Renal E. cuniculi disease tends to be progressive and clinically silent until significant renal mass has been lost. Azotemia in a rabbit without an obvious alternative explanation — urolithiasis, neoplasia, hypercalciuria — warrants E. cuniculi on the differential even in the absence of neurologic signs.
Phacoclastic uveitis presents as a white intraocular mass, anterior chamber inflammation, and variable pain in a typically young, otherwise healthy rabbit. It is the most pathologically specific presentation because the mechanism — lens capsule rupture and lens-induced uveitis in a young animal — is closely tied to the vertical transmission route. Enucleation with histopathology is both diagnostic and therapeutic and is the standard of care for affected eyes that do not respond to medical management.
Treatment and its limitations
Fenbendazole is the most widely used and best-supported treatment for E. cuniculi in rabbits, with controlled studies demonstrating efficacy in limiting spore shedding and reducing transmission. The standard protocol of 20 mg/kg orally once daily for 28 days is most commonly cited, though duration recommendations vary in the literature and longer courses are used in clinical practice. Fenbendazole does not eliminate established granulomatous lesions or reverse neurologic deficits — it targets the proliferative intracellular forms and reduces ongoing spore production. Established CNS fibrosis or significant renal scarring will not resolve regardless of treatment.
Supportive care — anti-inflammatory doses of meloxicam, nursing support, nutritional assistance — is as important as antiparasitic therapy in the acute phase, particularly for rabbits with vestibular disease severe enough to prevent normal ambulation and feeding. The prognosis for neurologic cases is variable; many rabbits show meaningful clinical improvement over weeks to months, and some residual deficits (persistent head tilt, mild ataxia) are compatible with a reasonable quality of life.
Postmortem diagnosis and what to submit
For rabbits dying with a history of neurologic, renal, or ocular disease where E. cuniculi has not been confirmed antemortem, necropsy with systematic tissue sampling is the most informative diagnostic step. Fixed tissues should include brain (multiple levels: cerebrum, cerebellum, brainstem), kidneys, eyes if affected, and spinal cord if signs suggest myelopathy. Fresh tissue for PCR — kidney and brain — is a valuable addition when available.
Inform the pathologist of the clinical history, serologic results if available, and the primary clinical presentation. The combination of characteristic histologic lesions, spore identification on special stains, and supportive PCR results provides the most defensible postmortem diagnosis. A report of granulomatous encephalitis and interstitial nephritis in the appropriate clinical context is strongly supportive even when spores are not directly identified — their small size and sparse distribution means a negative special stain does not exclude E. cuniculi.
A note on zoonotic potential
E. cuniculi is capable of infecting humans and has been documented as an opportunistic pathogen in immunocompromised individuals — particularly those with HIV/AIDS, transplant recipients, and others on immunosuppressive therapy. In immunocompetent individuals, infection is believed to be largely asymptomatic. The zoonotic risk from pet rabbits to healthy owners is considered low but not zero, and counseling immunocompromised rabbit owners on hygiene precautions — particularly around urine, which is the primary route of spore shedding — is appropriate and consistent with a conservative standard of care.
Eric Snook, DVM, PhD, DACVP — Vetopathy. Rabbit and exotic species histopathology is a particular area of interest. Necropsy submissions and antemortem biopsies welcome.