Beyond Lymphocytic-Plasmacytic IBD: Recognizing Eosinophilic Enteritis at Surgery
Some cases present as one problem and resolve as another. This is one of them. A young adult mixed-breed dog came to surgery for a small intestinal foreign body. What the biopsy revealed was a gut that had been inflamed long before the foreign body arrived.
Presentation and clinical context
The patient was a young adult mixed-breed dog with a prior surgical history — intestinal foreign body removal at approximately six months of age. The current presentation included episodic vomiting over recent weeks, progressive hyporexia over the preceding two to four weeks, a single episode of hematochezia with mucus, and increased mucoid coating on otherwise formed stools. Body condition score was 3.5/9.
The broader history was notable: the dog carried a longstanding diagnosis of chronic allergic disease. Owners reported constant pruritus, periorbital and ventral abdominal hair loss, and recurrent ear infections managed intermittently with diphenhydramine. A grain-free turkey and yam diet with commercial probiotics and allergy supplements had been in place for some time. The connection between the dermatologic history and the gastrointestinal presentation was not the presenting concern — it became relevant later.
Radiographs obtained by the referring veterinarian the day prior had identified a small intestinal foreign body with evidence of mechanical obstruction. Repeat films the following day showed persistent foreign material. POCUS at the time of surgical referral confirmed a large intraluminal foreign body, scant free peritoneal fluid, and — critically — generalized small intestinal wall thickening. That last finding was easy to attribute to obstruction-related edema. It warranted more careful consideration.
Clinicopathologic findings
Bloodwork revealed a mild non-regenerative anemia and a moderate inflammatory leukogram with mature neutrophilia — consistent with the obstructive and inflammatory process but not specific. The more diagnostically significant finding was a total protein of depressed to a level consistent with hypoalbuminemia at 2.0 g/dL. Lactate was within normal limits at 1.78 mmol/L, and pancreatic lipase was unremarkable.
Hypoalbuminemia in this context carried a differential list: protein-losing enteropathy from inflammatory bowel disease, hepatic synthetic failure, protein-losing nephropathy, or third-spacing from peritoneal effusion. With normal lactate and a modest free fluid finding on POCUS, hepatic and renal causes were less compelling than primary intestinal protein loss. The degree of hypoalbuminemia — below 2.0 g/dL — in a young dog with a chronic history of GI signs was a signal that something beyond obstruction was driving the clinical picture.
Intraoperative findings and the decision to biopsy
At surgery, the foreign body was identified and removed without enterotomy complication. The gross appearance of the intestinal wall at the surgical site was consistent with the edema and congestion expected from an obstructive event. There was no discrete mass, no obvious necrosis beyond what was attributable to the obstruction, and no gross finding that would have independently raised the index of suspicion for underlying mucosal disease.
Full-thickness intestinal biopsies were obtained from multiple sites — a decision that proves its value in exactly these cases. The gross intestine looked like a gut that had been obstructed. The histopathology told a different story.
Histopathologic findings
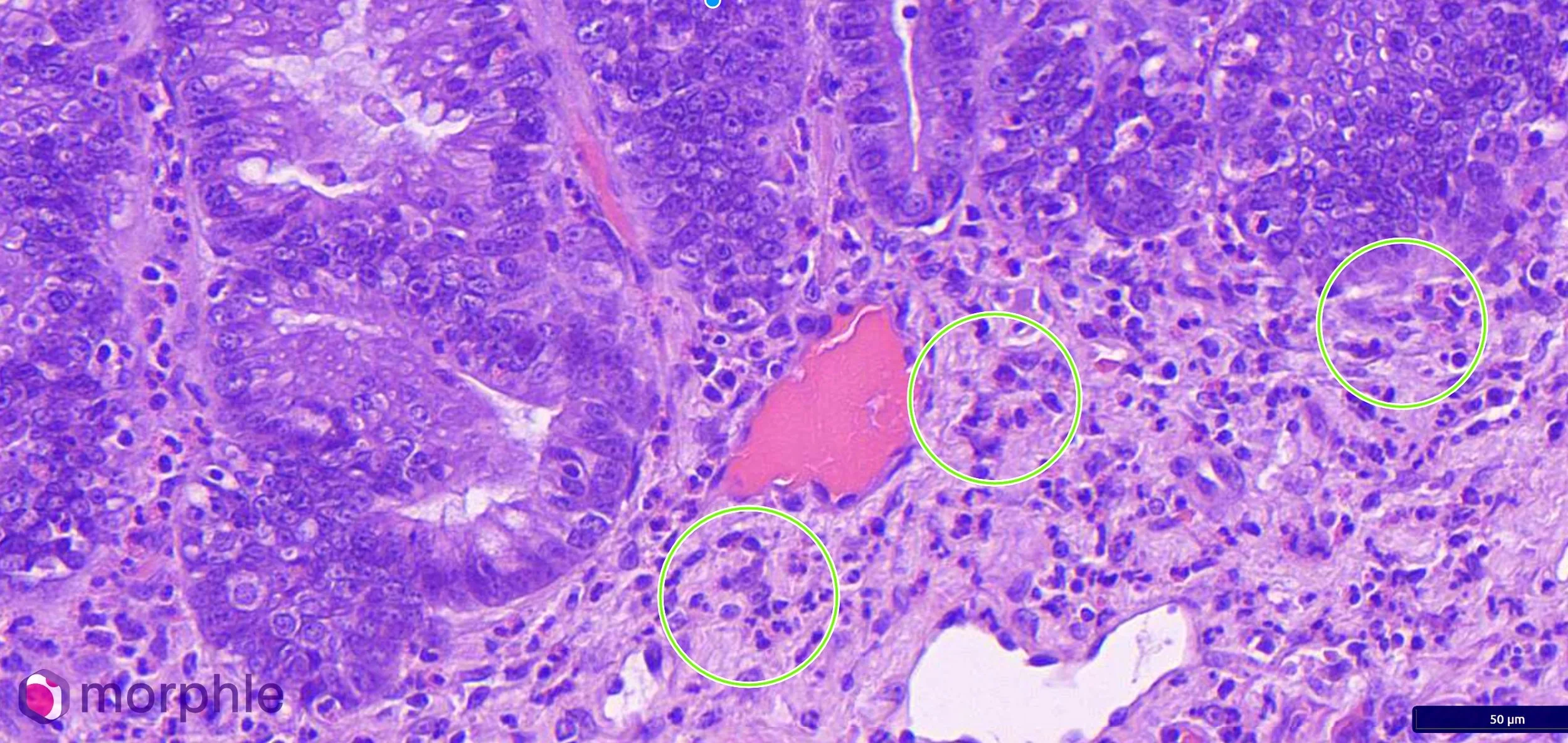
Sections of small intestinal wall revealed a marked eosinophilic infiltrate throughout the lamina propria. Eosinophils were present in high numbers in all sections examined, with infiltration extending into the crypts and disrupting the normal crypt architecture in areas. Villous blunting and crypt irregularity were present, consistent with chronic mucosal injury. The degree of eosinophilic infiltration was inconsistent with a reactive response to the foreign body alone — the distribution and severity indicated a pre-existing, active inflammatory process.
The diagnosis: eosinophilic enteritis. The foreign body had been the precipitating event that brought this dog to surgical attention. The underlying intestinal disease had been there first.
Histopathology of enteritis. The circles are representative areas with eosinophils present. Eosinophils are present in much higher numbers than typically expected in the instestine.
Eosinophilic enteritis: the pathologic and clinical picture
Eosinophilic enteritis is an inflammatory bowel disease subtype characterized by eosinophilic infiltration of the gastrointestinal mucosa, most commonly involving the small intestine. It is distinguished from other forms of chronic enteropathy by the predominance of eosinophils — a cell type associated with hypersensitivity responses, parasitic infestation, and mast cell-mediated immune activation. In dogs, it is frequently associated with food hypersensitivity or environmental allergic disease, though an underlying cause is not always identified.
The clinical presentation of eosinophilic enteritis overlaps substantially with other chronic enteropathies: chronic vomiting, hyporexia, weight loss, intermittent diarrhea, and — when protein-losing enteropathy develops — hypoalbuminemia. There is no gross endoscopic or surgical appearance that reliably distinguishes eosinophilic enteritis from lymphocytic-plasmacytic enteritis or other IBD subtypes. The mucosa may appear grossly normal, mildly thickened, or granular. The diagnosis is histopathologic.
In this case, the atopic history was not incidental. Dogs with systemic allergic disease — atopic dermatitis, environmental hypersensitivity, food allergy — have a demonstrably higher prevalence of concurrent gastrointestinal eosinophilic inflammation. Eosinophils are recruited to barrier tissues — skin, gut, respiratory mucosa — in response to allergen exposure and IL-5/IL-13-driven signaling. A dog with chronic pruritus, periorbital hair loss, and recurrent otitis is a dog with a dysregulated eosinophil-mediated immune response. It is not surprising that the gut was involved. The surprise is that it took a foreign body to find it. The finding of eosinophilic enteritis also raises the question of whether this dog’s dermatologic signs may be partly or wholly food-driven rather than purely environmental. Food hypersensitivity can produce a concurrent cutaneous and gastrointestinal eosinophilic response, with the gut and the skin serving as parallel target organs for the same underlying immune dysregulation. Simultaneous gastrointestinal and dermatologic signs have been reported in 20 to 83% of dogs with adverse food reactions, including vomiting, diarrhea, flatulence, and borborygmus alongside cutaneous signs (de Farias et al., Royal Canin Academy). Separately, biomarker studies in dogs with atopic dermatitis have identified subclinical intestinal mucosal barrier damage even in the absence of overt GI signs, suggesting that gut involvement in the atopic dog is more common than clinical presentation alone would indicate (Gulersoy et al., J Vet Res, 2024). Food allergy in dogs should be considered as a differential whenever pruritus, recurrent otitis, and gastrointestinal signs co-occur and other causes have been excluded (Verlinden et al., JAVMA, 2023). The current grain-free turkey and yam diet does not exclude food antigen exposure as a driver — novel protein diets are not synonymous with hydrolyzed protein diets, and turkey itself can be a sensitizing antigen in a dog with a prior exposure history. Distinguishing food hypersensitivity from environmental atopy as the primary driver of this dog’s allergic phenotype requires a strict dietary elimination trial with a hydrolyzed or truly novel protein source, conducted for a minimum of eight to twelve weeks. That step was not possible to complete prior to surgery but warrants prioritization in the post-operative management plan.
Hypoalbuminemia as a diagnostic signal
The albumin of 2.0 g/dL in this case deserves emphasis. Hypoalbuminemia in a young dog with chronic GI signs is not a nonspecific finding to be noted and filed — it is a marker of meaningful protein loss from the gastrointestinal tract, reduced hepatic synthesis, or both. In this context, with a chronic history that preceded the acute obstruction, protein-losing enteropathy from ongoing mucosal inflammation was the most parsimonious explanation.
Eosinophilic enteritis can produce clinically significant protein-losing enteropathy when mucosal infiltration is extensive enough to impair absorptive surface area and increase mucosal permeability. The albumin level in this case was consistent with that mechanism. It also carried prognostic and therapeutic implications — hypoalbuminemia influences wound healing, drug pharmacokinetics, oncotic pressure, and anesthetic risk. The ASA II designation assigned at the time of surgery was appropriate given the clinical stability, but the hypoalbuminemia underscored that this was not a routine obstruction.
Clinical takeaway
Intraoperative gross assessment of the bowel is not a substitute for histopathology. An intestinal wall that has been obstructed looks inflamed. An intestinal wall with pre-existing eosinophilic enteritis also looks inflamed. Without tissue examination, the two are indistinguishable — and the distinction matters for the conversation that follows surgery.
In any dog undergoing intestinal surgery for foreign body, chronic hyporexia, weight loss, or hypoalbuminemia, full-thickness biopsy from multiple sites should be routine. The incremental procedural cost and operative time are minimal. The diagnostic return — particularly in cases where an underlying inflammatory enteropathy has been operating subclinically — is substantial.
This dog's GI disease did not start the day the foreign body was ingested. The history of chronic allergic disease, progressive hyporexia, mucoid stool, and hypoalbuminemia outlined a trajectory that had been building for months. The biopsy made it visible.
References
de Farias et al. Adverse Food Reactions in Dogs: what do we currently know? Royal Canin Veterinary Academy. Available at: academy.royalcanin.com. [Reports 20–83% co-occurrence of GI and cutaneous signs in dogs with adverse food reactions.]
Gulersoy E et al. Investigation of the relationship between atopic dermatitis of dogs and intestinal epithelial damage. J Vet Res. 2024; PMC11034634. [Elevated intestinal barrier repair biomarkers in dogs with atopic dermatitis indicate subclinical chronic intestinal mucosal damage.]
Verlinden A et al. Food allergy in dogs and cats: current perspectives on etiology, diagnosis, and management. J Am Vet Med Assoc. 2023;261(S1). doi:10.2460/javma.22.12.0548. [Concurrent pruritus, recurrent otitis, and GI signs warrant food allergy as a differential; food allergy occurs in up to 50% of atopic dermatitis cases.]
Case submitted to Vetopathy. Diagnosis rendered by Eric Snook, DVM, PhD, DACVP. All patient details anonymized.